
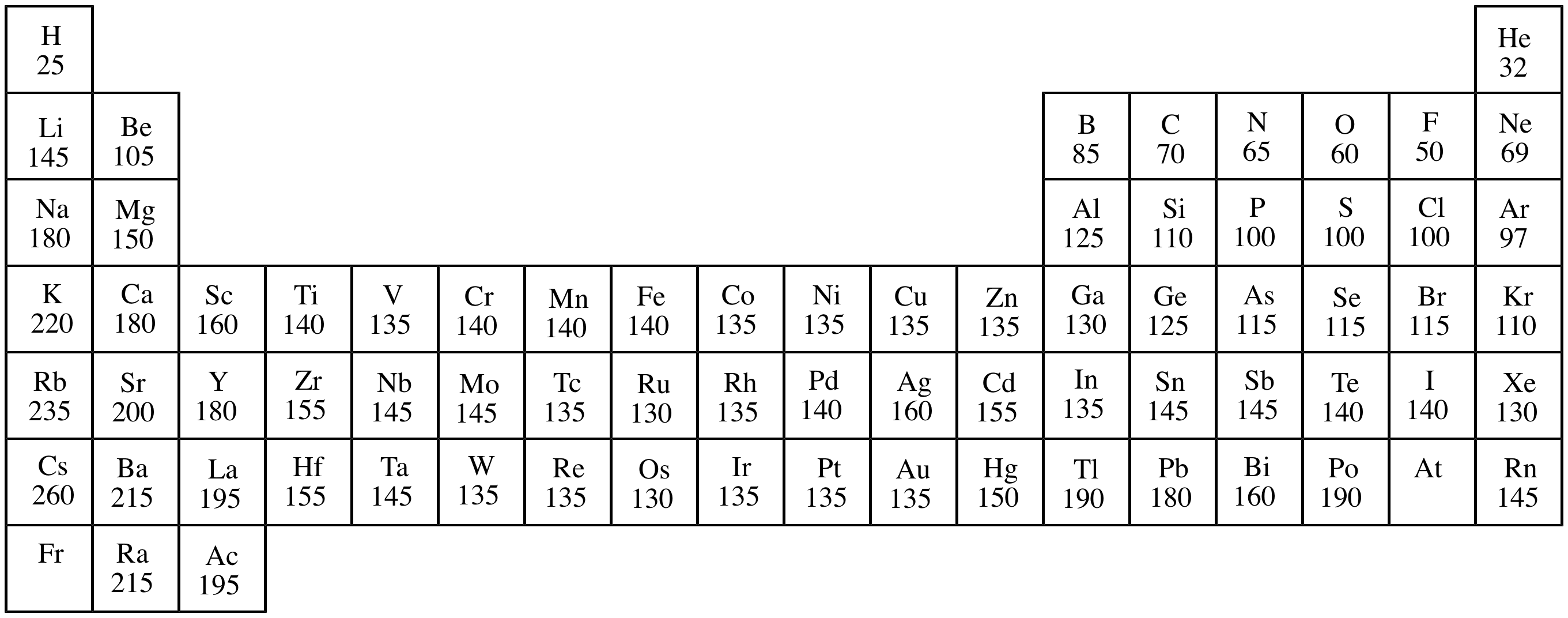
Figure 4: The ionic radius increases for the generation of negative ions. The gain of an electron adds more electrons to the outermost shell which increases the radius because there are now more electrons further away from the nucleus and there are more electrons to pull towards the nucleus so the pull becomes slightly weaker than of the neutral atom and causes an increase in atomic radius. Figure 3: The ionic radius decreases for the generation of positive ions.Īn anion, on the other hand, will be bigger in size than that of the atom it was made from because of a gain of an electron. Thus, helium is the smallest element, and francium is the largest. As can be seen in the figures below, the atomic radius increases from top to bottom in a group, and decreases from left to right across a period. 
A neutral atom X is shown here to have a bond length of 180 pm and then the cation X + is smaller with a bond length of 100 pm. Atomic radii vary in a predictable way across the periodic table. This can similarly be said about the protons pulling the electrons closer to the nucleus, which as a result decreases atomic size.įigure 3 below depicts this process. If ten magnets and ten metallic objects represent a neutral atom where the magnets are protons and the metallic objects are electrons, then removing one metallic object, which is like removing an electron, will cause the magnet to pull the metallic objects closer because of a decrease in number of the metallic objects.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
